Physicists in West Virginia have announced a potential breakthrough that could help upend a longstanding constraint imposed by the first law of thermodynamics.
The discovery, involving how energy is converted in plasmas in space, was described in new research published in the journal Physical Review Letters, and could potentially require scientists to have to rethink how plasmas are heated both in the lab and in space.
The first law of thermodynamics, an expression of the law of conservation of energy albeit styled with relation to thermodynamic processes, conveys that the total energy within a system will remain constant, but that it can be converted from one form of energy into another. More simply, the idea is commonly expressed as “energy can neither be created or destroyed.”
“The first law has been used to describe many things,” says Paul Cassak, a professor of Theoretical and Computational Plasma Physics at West Virginia University’s Department of Physics and Astronomy. Cassak is associate director of the Center for KINETIC Plasma Physics, where along with graduate research assistant Hasan Barbhuiya he studies the ways energy is converted in superheated plasmas in space.

“The laws of thermodynamics were developed about 170 years ago,” Cassak told The Debrief, “and the technology of the time dictated the gases or fluids that people would have studied are in equilibrium at the densities and temperatures that they were using back then.”
A good example of equilibrium would be how individual vessels filled with water at different temperatures within the same environment will eventually either cool or warm until they reach the same temperature. However, the conditions under which the laws of thermodynamics were developed at that time dictated that they would onlywork for systems in equilibrium, and Cassak says attempting to revise existing theories about thermodynamics hasn’t been a simple task.
“The part that has been challenging has been the theoretical work to figure out how to extend or improve upon the original laws of thermodynamics when systems aren’t in equilibrium,” Cassak told The Debrief.
“People have been working on extending the laws of thermodynamics for systems not in equilibrium for over 100 years,” he says. “Most of what people had figured out is how to extend the laws of thermodynamics for systems that are close to equilibrium. Even this is a huge step forward and has been useful in many fields of science.”
In certain settings (like with space plasmas), systems can be far from being near equilibrium, so the approximations do not work. Hence, explaining the energy conversion is more challenging.
However, Cassak and Barbhuiya say they devised a way to generalize the first law of thermodynamics, which allows it to be applied to systems that are not in equilibrium.
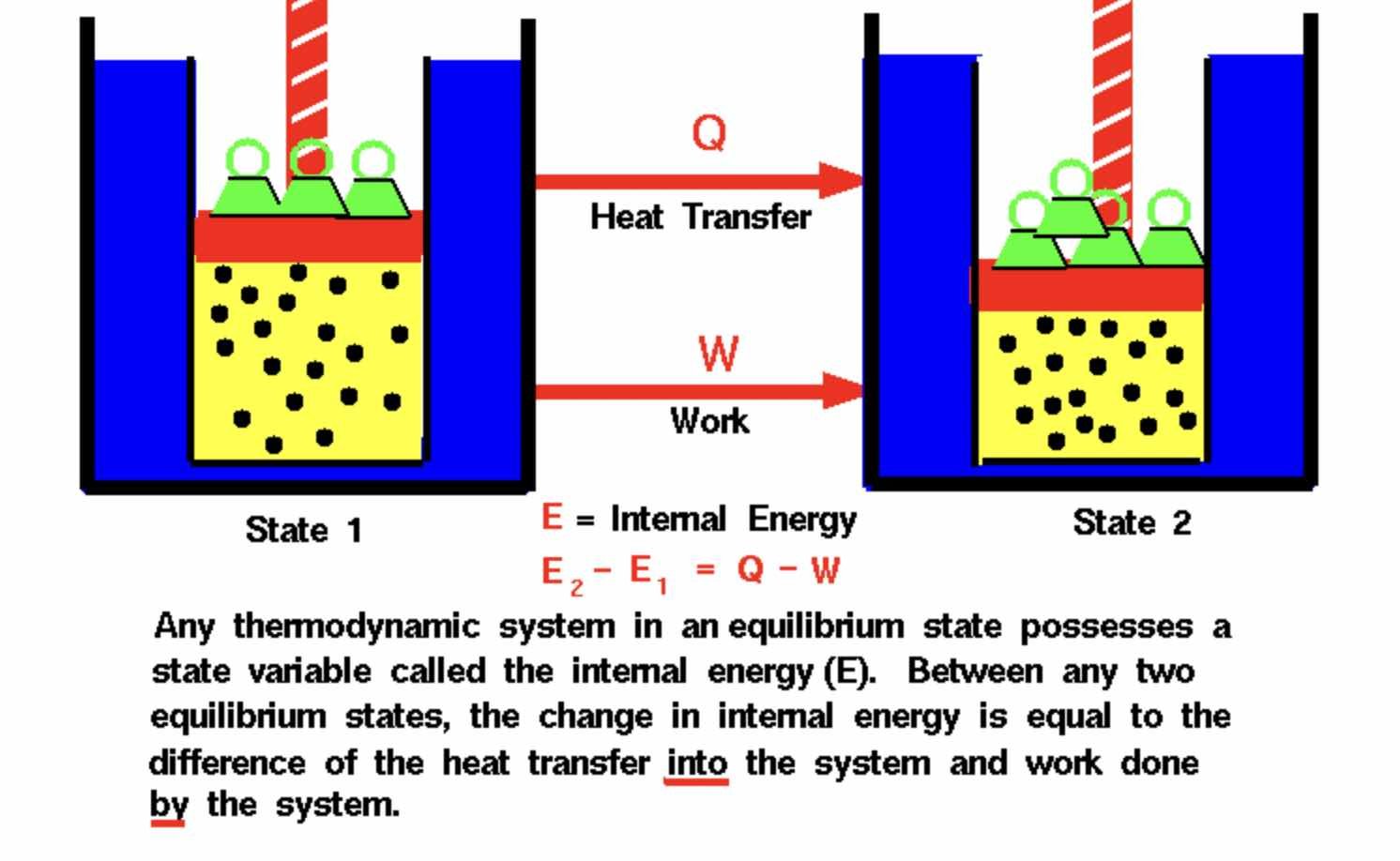
According to Cassak, there are three main points involved, the first being that for a simple, “ideal” gas, liquid, or plasma in equilibrium, its properties can be described by two main quantities: density, and temperature.
“The first law of thermodynamics, then, describes energy conversion for a process that changes the density and/or temperature,” Cassak told The Debrief.
“The second key point is that, for a gas, liquid, or plasma that is not in equilibrium, its properties can no longer be described by only the density and temperature,” he adds. Generally, there are almost an infinite number of different quantities that are needed to describe a substance under such conditions.
“So, one still has the density and temperature, but also many many more quantities,” Cassak says.
The third point involves how a system, when out of equilibrium, continues to be described by the first law of thermodynamics. Cassak says that “when a system is out of equilibrium, the first law of thermodynamics continues to only describe energy conversion that changes the density and temperature.”
“We realized that the energy conversion associated with all the other quantities describing the gas, liquid, plasma, etc., when it is not in equilibrium are not included in the first law of thermodynamics,” Cassak told The Debrief. To overcome this, he and Barbhuiya “derived an equation that quantifies not just the energy conversion that changes the density and temperature, but also all of these other quantities that describe the system when it’s not in equilibrium.”
Previously, state-of-the-art research in this area could account for energy conversion only with relation to expansion and heating. However, the team’s newly generalized theory offers a way to now calculate all of the energy from not being in equilibrium.

Cassak characterized the team’s results as “a really large step in our understanding,” and their findings also have several promising applications, which includes allowing scientists studying plasmas in space to better understand their dynamics. Namely, this could enhance our understanding of space weather events like mass coronal ejections from the sun that release large plumes of superheated plasma capable of interfering with communications systems and other technologies here on Earth.
“Because the first law of thermodynamics is so widely used, it is our hope that scientists in a wide array of fields could use our result,” Barbhuiya added in a statement.
Duncan Lorimer, a professor of astronomy at West Virginia University best known for having been the first to discover a fast radio burst in 2007, offered praise for Cassak and Barbhuiya’s work.
“To take one of these laws that has been around over 150 years and improve on it is a major achievement,” Lorimer said in a statement.
The paper, “Quantifying Energy Conversion in Higher-Order Phase Space Density Moments in Plasmas,” by Cassak, Barbhuiya, et al, appeared in Physical Review Letters on February 22, 2022.
Micah Hanks is the Editor-in-Chief and Co-Founder of The Debrief. He can be reached by email at micah@thedebrief.org. Follow his work at micahhanks.com and on Twitter: @MicahHanks.

